Have any Patents Issued under the USPTO’s COVID-19 Prioritized Patent Examination Program?
In a previous IP Alert, we reported that the U.S. Patent and Trademark Office (USPTO) introduced the COVID-19 Prioritized Examination Pilot Program to accelerate examination of certain patent applications directed to products or processes related to the treatment or prevention of COVID-19. Qualifying applicants will have official USPTO application fees waived and can expect final disposition of their patent application within 6-12 months.
The program was initially limited to 500 patent applications, but the USPTO indicated that the number could be increased depending on the program effectiveness and its impact on USPTO resources.
As of January 19, 2021, 474 requests under the program had been filed with the USPTO, and 278 requests had been granted, thus leaving 222 remaining grants available. More than half of the applications granted prioritized examination were directed to medical treatments, vaccines, and diagnostic technology, while the remainder were directed to ventilators, personal protective equipment, and other technology related to COVID-19.
Patents Issued under the COVID-19 Prioritized Examination Program
On December 28, 2020, the USPTO announced that 33 of the applications accepted into the program were either allowed or granted. The following is a brief summary of a few of the patents issued under the program:
U.S. Patent No. 10,842,697 – “Disposable Bio-Secure Environmental Unit”
This patent is directed to a disposable, bio-secure enclosure designed to be attached to hospital stretchers and beds for isolating and transporting patients suspected of having an infectious or contagious disease. As shown in FIG. 1 of the patent, the enclosure can also include a number of gloved access points that allow practitioners to interact with the patient, while also allowing mobility for the patient to sit up and move their arms and legs.

FIG. 1 of U.S. Patent No. 10,842,697
U.S. Patent No. 10,874,687 – “Highly Active Compounds against COVID-19”
This patent is directed to COVID-19 drug treatments and, more particularly, to purine nucleotide antivirals that inhibit replication for a number of RNA viruses, including coronaviruses and flaviviruses. The purine nucleotides target viral RNA polymerase, a key enzyme used to produce copies of the virus’ RNA genome, halting the virus life cycle and the production of new viruses.
U.S. Patent No. 10,803,714 – “System and Method of Visualizing, Tracking, and Maintaining Social Distancing”
This patent is directed to limiting viral spread by promoting social distancing. As shown in FIG. 3B of the patent, a visible boundary generating device can be worn by a user and is designed to produce a 360° zone of visible, colored light encircling the user. The visible light represents a safe zone and has a radius conforming to specified social distance parameters. The system can also be extended to mobile device applications for recording location data and providing proximity notifications or alerts for “social distancing incursions.”
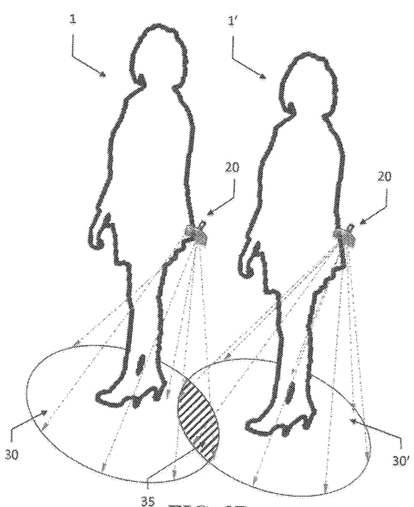
FIG. 3B of U.S. Patent No. 10,803,714
U.S. Patent No. 10,834,978 – “Soft Silicone Edged Cushion for Face Masks”
This patent is directed to a silicone cushion that can be attached to face masks to reduce air leakage and provide comfort to its wearer. In one example, the cushion can be U-shaped and clasped to the lateral edges of the face mask. The cushion can also contain a silicone gel that enhances user comfort and leak resistance.
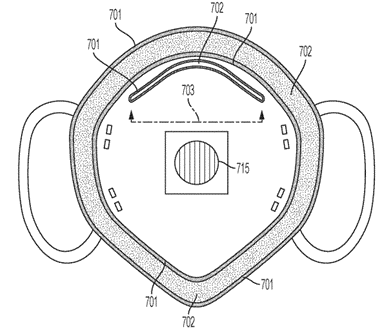
FIG. 7 of U.S. Patent No. 10, 834,714
Conclusion
The COVID-19 Prioritized Examination Pilot Program remains in effect and the USPTO has not indicated whether the number of requests will be extended beyond the initial 500 at this time. Please reach out to the patent attorneys at Vorys with any questions or for assistance in applying for the program.

